Reasons for appearance
To date, scientists cannot say for sure what provokes the appearance of this disease.
There is evidence that the irritating factor is a viral infectious process (influenza, viral hepatitis type A). The disease can also be provoked by taking drugs such as Carbimazole and Penicillamine. Other causes of the disease include inhalation of harmful substances, hypothermia and smoking. Therefore, it is very important to monitor your health and lifestyle
The disease can be transmitted genetically. The medical literature describes cases where the syndrome passed on to relatives. This disease can occur suddenly due to changes in the body's immune system. Under the influence of aggressive factors, the body begins to produce antibodies to structures such as basement membranes in the pulmonary arterioles and renal glomeruli.
It is generally accepted that in the bloodstream the role of an antigen is played by a component such as the a-3 chain of type IV collagen. It is found in the kidney capillaries and basement membrane structures. Antibodies, together with C3 complement, combine with antigens, and then the resulting immune complexes accumulate along the basement membrane. They induce inflammation that affects the renal glomerular structures. As a result of such changes, disease occurs in the form of alveolitis and glomerulonephritis.
In the development of the first variant of the disease in Goodpasture syndrome, the irritating factor is considered to be the process of activation of macrophages, which in this condition release cytokines (about 40). The first group includes the following representatives: inteleikin-8, chematoxins, leukotrienes. Their task comes down to enhancing the process of passage of polymorphonuclear leukocytes into the lungs.
And group 2 cytokines help move fibroblasts to this organ. Among the causes of their growth are macrophage and platelet. In addition, alveolar macrophages are capable of producing proteases, reactive oxygen species that damage lung tissue.
Diagnostics
The diagnosis of anti-GBM disease may be suspected based on the identification of characteristic physical signs (eg, pulmonary hemorrhage and glomerulonephritis). The diagnosis can be confirmed by identifying the presence of antiglomerular basement membrane antibodies in the body. In some cases, affected people may have blood (hematuria) and/or protein (proteinuria) in the urine.
What causes Goodpasture syndrome?
The cause of the disease has not been precisely established. A genetic predisposition to Goodpasture syndrome is assumed; the presence of HLA-DRW2 is considered its marker. There is a point of view about the possible role of a previous viral infection (hepatitis A virus and other viral diseases), industrial hazards, and medications (primarily D-penicillamine).
The basis of the pathogenesis of Goodpasture syndrome is the formation of autoantibodies to the basement membranes of the capillaries of the glomeruli and alveoli. These antibodies belong to the IgG class; they bind to basement membrane antibodies in the presence of the C3 component of complement, with the subsequent development of immune inflammation of the kidneys and alveoli of the lungs.
Anti-GBM antibodies are directed against the non-collagenous (NC-1) domain of the a3 chain of type IV collagen, which is found in highest concentration in the basement membranes of renal and pulmonary capillaries. Exposure to environmental factors - smoking, viral acute respiratory infections and inhalation of bicarbonate suspensions (more often) - and, less often, pneumonia activates the presentation of alveolar capillary antigens to circulating antibodies in people with a hereditary predisposition (most often these are carriers of the HLA-DRwl5, - DR4 and - DRB1 alleles ). Circulating anti-GBM antibodies bind to basement membranes, fix complement, and cause a cellular inflammatory response, leading to the development of glomerulonephritis and/or pulmonary capillaritis.
There is probably a certain commonality of auto-antigens in the basement membrane of the capillaries of the glomeruli and alveoli. The autoantigen is formed under the influence of the damaging effect of the etiological factor. An unknown etiological factor damages and modifies the structure of the basement membranes of the kidneys and lungs. The excretion of the resulting degradation products of the glomerular basement membranes of the kidneys when they are damaged slows down and decreases, which naturally creates the preconditions for the development of autoimmune damage to the kidneys and lungs. It is still not completely known which component of the basement membrane becomes an auto-antigen. It is currently believed that this is an internal structural component of the glomerular basement membrane of the kidney, the α3 chain of type 4 collagen.
The formed immune complexes are deposited along the basement membranes of the glomerular capillaries, which leads to the development of an immunoinflammatory process in the renal glomerulus (glomerulonephritis) and alveoli (alveolitis). The main cells involved in the development of this immune inflammation are T lymphocytes, monocytes, endothelial cells, polymorphonuclear leukocytes, and alveolar macrophages. The interaction between them is ensured by molecular mediators, cytokines (growth factors - platelet, insulin-like, b-transforming; interleukin-1, tumor necrosis factor, etc.). Metabolites of arachidonic acid, free oxygen radicals, proteolytic enzymes, and adhesive molecules play a major role in the development of immune inflammation.
Factors and mechanism of development of pathology
The reasons for the development of this syndrome are little known, but there are a number of etiological factors:
- taking medications from a number of immunosuppressants (Azathioprine, Penicillamine);
- some genetic predisposition;
- past viral infections (ARVI);
- inhalation of volatile hydrocarbons, exhaust gases, petroleum products;
- smoking, including drugs;
- inhalation therapy with oxygen in high concentrations (oxygen therapy).
The main mechanism for the development of pulmonary-renal syndrome is a failure in the central organs of the immune system, possibly leading to a violation of the negative selection of lymphocytes. This subsequently leads to the appearance of a pathological clone of B-lymphocytes, which produces auto-aggressive antibodies. Further, with Goodpasture's syndrome, according to the cascade model, autoimmune processes are launched, leading to damage to connective tissue, mainly the capillaries of the kidneys and alveoli of the human respiratory organ, which is expressed in persistent dysfunction of these systems.
Symptoms of Goodpasture's syndrome
The disease most often begins with clinical manifestations of pulmonary pathology. Hemoptysis is the most noticeable symptom; however, hemoptysis may be absent in the presence of hemorrhagic manifestations, and the patient may show only infiltrative changes on chest x-ray or infiltrate and respiratory distress and/or failure. Shortness of breath (mainly during exercise), cough, malaise, decreased ability to work, chest pain, fever and weight loss often develop. Up to 40% of patients have gross hematuria, although pulmonary hemorrhage may precede renal manifestations by weeks to years.
During hemoptysis, shortness of breath may increase. Weakness and decreased ability to work are also concerning.
Symptoms of Goodpasture syndrome vary over time, ranging from clear lungs on auscultation to crackling and dry rales. Some patients experience peripheral edema and pallor due to anemia.
Upon examination, attention is drawn to pallor of the skin, cyanosis of the mucous membranes, pastiness or severe swelling of the face, decreased muscle strength, and loss of body weight. Body temperature is usually elevated to febrile levels
When percussing the lungs, a shortening of the percussion sound can be detected over extensive foci of pulmonary hemorrhages, but this is rarely observed; more often there are no changes in the percussion sound.
A characteristic auscultatory sign of Goodpasture's syndrome is dry and moist rales, their number increases significantly during or after hemoptysis.
When examining the cardiovascular system, arterial hypertension is revealed, the border of relative dullness of the heart may increase to the left, muffled heart sounds, a quiet systolic murmur; with the development of severe renal failure, a pericardial friction noise appears. With progressive kidney damage against the background of significant arterial hypertension, the development of acute left ventricular failure with a picture of cardiac asthma and pulmonary edema is possible. Usually this situation develops in the terminal stage of the disease.
As a rule, kidney damage appears later, after a certain time after the development of pulmonary symptoms. Characteristic clinical signs of renal pathology are hematuria (sometimes gross hematuria), rapidly progressing renal failure, oligoanuria, and arterial hypertension.
In 10-15% of cases, Goodpasture syndrome begins with clinical signs of renal pathology - a clinical picture of glomerulonephritis appears (oliguria, edema, arterial hypertension, severe pallor), and then symptoms of lung damage appear. Many patients may have myalgia and arthralgia.
Symptoms
At the onset of the disease, nonspecific symptoms of Goodpasture syndrome appear:
- malaise, unexplained weakness, fatigue,
- increased body temperature for no clear reason,
- pain in various joints without swelling or redness,
- weight loss.
Already at this stage, even in the absence of pulmonary bleeding, the patient develops anemia.
Nonspecific symptoms in Goodpasture syndrome are usually less pronounced than in other rheumatological diseases. Often the patient does not see a doctor, and the disease begins to progress rapidly.
In almost 70% of patients, the first symptom of the disease is hemoptysis. This is especially true for smokers. Now doctors are noticing a decrease in the frequency of this symptom, which may be due to a decrease in the prevalence of this bad habit.
In addition to hemoptysis, symptoms include shortness of breath and cough, and later pulmonary hemorrhage develops.
Pulmonary hemorrhage does not necessarily occur against the background of hemoptysis and does not depend on its severity. In many patients it develops suddenly and causes death within a few hours. This condition is accompanied by severe shortness of breath and bluish skin. It may be complicated by pulmonary edema or pneumonia.
A few months after the onset of the first pulmonary symptoms, a patient with Goodpasture syndrome develops rapidly progressive glomerulonephritis, which causes renal failure. This condition also often leads to death within the next two years.
The main symptoms of kidney damage:
- weakness, lack of appetite, pale skin,
- headache and lower back pain,
- various pains in the heart area,
- increased blood pressure,
- widespread swelling,
- blurred vision,
- decrease in urine volume.
These symptoms of Goodpasture syndrome fully manifest themselves within 4 to 6 weeks after their onset. Therefore, such glomerulonephritis is called rapidly progressive.
Mechanism of capillary damage
from the arterial part, the brought nutrients and oxygen are released into the tissue cells, the venous part takes waste materials and removes them to the cleansing organs (liver and kidneys).
Normally, up to half of the capillaries are in a collapsed state and do not work. They provide a “reserve” in case of failure of existing vessels and increased tissue demand. Capillaries are found in all organs and tissues. They form different forms of tortuosity: from a “hook” to a “ball” (inside the kidneys).
The basement membrane also consists of three layers; they are formed by collagen filaments, protein-carbohydrate and protein-fat complexes (glycoproteins and lipoproteins)
The most important function is filtration, the one-way passage of certain substances through the wall. Capillaries control the retention of blood cells, protein, and necessary plastic materials. For this purpose, a three-layer barrier is formed in the structure of the wall:
- endothelium from the inside,
- in the middle - the basement membrane,
- outwardly - a layer of surface cells (podocytes or pericytes), with their processes grown into the membrane; it is to this type of cells that nerve endings are suitable, which can cause spasm or dilation of blood vessels.
The pathogenesis of Goodspacher's syndrome is associated with selective inflammation of the basement membrane in the capillaries of the alveoli of the lung tissue and in the renal glomeruli. The inflammatory reaction is autoimmune in nature.
“Self-defense” provided by immune cells significantly exceeds the need and does not stop, causing damage to the body. It has been established that destructive antibodies have an affinity for a certain type of collagen, which is located in maximum quantities in the kidneys and alveoli. How this commonality is established is not yet known.
Under the influence of the above reasons, the cells of the basement membrane begin to be perceived as foreign, and antibodies of the GBM type are produced against them. Fixation of complexes with antibodies occurs along the membrane, which causes inflammation (capillaritis), and in the kidneys - glomerulonephritis.
Accomplices of the hyperreaction are blood cells and tissue elements
Proven role in inflammation:
- T-lymphocytes,
- monocytes,
- polymorphonuclear leukocytes,
- endothelial cells,
- alveolar macrophages - produce 40 types of cytokines, a protease enzyme that damages the alveoli of the lungs.
The general interaction is supported by cytokines, which include:
- growth factors (including platelet, b-transforming, insulin-like);
- interleukin-1;
- tumor necrosis factor.
The reaction mechanism in the body is provided by:
- breakdown products of arachidonic acid;
- free radicals of oxygen molecules;
- proteolytic enzymes;
- adhesion molecules (molecular combinations that promote the attachment of one immune cell to another to ensure their movement through the bloodstream).
Mechanism of disease development
Goodpasture syndrome is an autoimmune disease, i.e. caused by the generation of antibodies to one’s own cells. The glomerular basement membrane is where these antibodies are produced. These antibodies bind to a specific domain within the fourth type of collagen.
It is this part of the fourth type collagen that is the target point of antibodies. This portion of type 4 collagen is called Goodpasture antigen.
In healthy people, this antigen is not a trigger for pathological chains. The disease affects lung and kidney tissue, because these tissues contain a large number of antigens of this type.
When antibodies form a bond with the Goodpasture antigen, the complement system is launched. These are immune proteins, or rather their special type. This formed bond is the trigger for a pathological protein chain reaction. At the point of contact of antibodies and antigens, communication with leukocytes occurs.
Everything goes to the point that leukocytes actively attack the affected areas of tissue, thereby destroying them. The immune response to this process is a sharp increase in the number of epithelial cells. Their significant deposition occurs on the surface of the basement membrane. The organ begins to quickly lose its functionality, cannot cope with the load, and waste products quickly accumulate in the body.
The mechanisms of pathological changes in the kidneys and lungs have an identical course.
Treatment
If there are no signs of pulmonary bleeding, oral hormones are started immediately. According to studies, the effect of taking hormonal drugs in combination with immunosuppressants - cyclophosphamide or azathioprine - increases.
The following medications are used to treat the disease:
- Prednisolone.
- Indomethacin.
- Chimes.
- Azathioprine.
In 1976, Mukhin N.A proposed an algorithm for the treatment of Goodpasture syndrome. Used:
- Prednisolone 1 mg/kg per day;
- Cyclophosphamide 3 mg/kg per day;
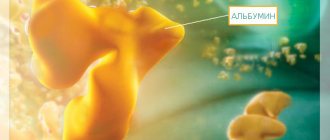
- Plasmapheresis – sessions are carried out daily for 2 weeks. 4 liters of blood plasma is changed to a 5% albumin solution, as well as fresh frozen plasma of the same blood group. If pulmonary bleeding is present, an additional 300-400 ml of fresh frozen plasma is administered.
After the main treatment, maintenance treatment is prescribed with reduced doses of the main drugs.
It is proposed to subsequently resolve the issue of using immunoadsorption and monotherapy with cyclosporine A.
The disease can be detected using x-rays. The radiograph will show small focal changes and deformation of the pulmonary pattern. Changes most often occur in the lower lobes. A detailed interview with the patient is important. This will help determine the exact cause of the syndrome and begin treatment.
In addition to X-rays, urine and blood tests are performed: the results will help to recreate the exact picture of the syndrome:
- If the kidneys are damaged, blood and protein will be seen in the urine.
- Antibodies to glomerular basement membranes can be detected in the blood.
- If necessary, a liver biopsy is performed - tissue samples can reveal changes characteristic of the syndrome.
- When examining sputum, siderophage cells can be identified.
You need to do a general blood test
Also used:
- cytostatics;
- prednisolone is a steroid hormone that suppresses inflammation;
- cyclophosphamide, which interferes with the formation of autoantibodies.
In advanced cases, the kidney is removed followed by transplantation, but, as was said, this procedure does not always protect against relapse. If the patient continues to lead an unhealthy lifestyle or remains in the place where the respiratory tract has become infected, the disease will quickly progress in the transplanted kidney. Cases of spontaneous remission have been observed even in the absence of infection factors.
The prognosis of Goodpasture syndrome is not always favorable - if the disease was detected in the early stages, that is, at the time of hemoptysis, then the survival rate increases significantly. If the situation progresses to renal or pulmonary failure, then death is possible in {amp}gt;50% of cases.
Treatment of Goodpasture syndrome comes down to excluding developing complications and their prevention
Providing emergency care to patients is of no small importance. In the acute period of the syndrome, pulse therapy is used:
- Methylprednisolone;
- Cyclophosphamide along with Methylprednisolone.
Symptomatic treatment of the syndrome includes:
- plasmapheresis;
- transfusion of red blood cells;
- use of iron supplements.
The course of the disease in most cases is steadily progressive, so the prognosis is not very encouraging. The diagnosis of Goodpasture syndrome in most cases is a death sentence. The death of patients occurs due to profuse pulmonary hemorrhages, renal failure, and respiratory failure.
In the malignant form of the disease, death occurs within a few weeks. In other cases of the disease, life expectancy is on average 1–3 years. Cases of remission of the disease Goodpasture syndrome are rare and practically not described.
Diagnostic and observation methods
The diagnosis is made on the basis of a serological analysis to detect serum antibodies in the blood. In order to differentiate diseases with similar clinical manifestations, such as hemorrhagic vasculitis, systemic lupus erythematosus and Wegener's syndrome, Goodpasture syndrome is diagnosed using the following methods:
- blood and urine tests;
- checking the main indicators of the immune system (immunogram);
- lung and kidney biopsy;
- electrocardiography;
- plain radiography;
- Ultrasound of the kidneys;
- measurement of respiratory parameters;
- general sputum analysis.
Kidney ultrasound
The main laboratory markers of Goodpasture syndrome are: high titer of antibodies to blood serum (up to 1000 U/ml and above) in enzyme immunoassay and histology results in kidney biopsy.
In case of suspicion and further examination for Goodpasture syndrome, it is necessary to conduct an examination of the initial state of the immune system with the determination of the main cell populations.
When the diagnosis is confirmed, a monitoring study is carried out for further therapy and observation. The fundamental ones here are:
- Determination of the titer of antibodies to the basement membrane of the glomeruli of the kidneys. It is carried out once every 2 weeks after a course of immunosuppressive therapy.
- A general blood test is prescribed from several times a day to 1 time a week.
- A general urine test is prescribed daily or weekly depending on the clinical picture.
- A biochemical blood test to determine urea and creatinine is carried out according to indications.
Analysis for urea determination
Further studies of the immune status are prescribed during therapy. If the patient reaches a stable level of recovery, he is scheduled for follow-up every 3, 6, 12 months. Unfortunately, patients with Goodpasture syndrome die within the first year of diagnosis. According to the observations of specialists, patients with remission underwent a course of specific immunosuppressive treatment.
Reasons for development
The etiology of the disease still remains unclear. It is believed that the pathological process is based on immunological damage: antibodies are produced to the basement membranes of the renal glomeruli and pulmonary alveoli, which lead to inflammation and disruption of the kidneys and lungs. The combined damage to these organs is explained by the common antigenic structure of their basement membranes.
It is not uncommon for the syndrome to occur shortly after influenza or inhalation of hydrocarbons. In this regard, it is assumed that such influences change the chemical structure of membrane antigens. As a result, they acquire autoantigenic properties, causing the production of antibodies. Thus, regarding the reasons for the formation of autoantibodies, there are several hypotheses:
- infectious diseases or toxic effects of chemicals;
- the action of exogenous (entered the body from the environment) antigens;
- slowing down the excretion of breakdown products of basement membranes due to impaired excretory function of the kidneys.
Some doctors are inclined to believe that Goodpasture syndrome is a form of idiopathic pulmonary hemosiderosis. The hypothesis about the viral nature of the disease has not been confirmed to date.
Under normal conditions, the endothelium is a barrier to antibodies. But when there is an increase in the permeability of blood vessels, their deposition on the basement membrane begins. Since the human body is a closed system and all processes are interconnected, the factors that cause damage to the lungs will then inevitably cause damage to the kidneys. Thus, the reason for the increase in capillary permeability can be:
- increase in capillary hydrostatic pressure;
- high concentrations of oxygen in the inhaled air;
- inhalation of metal dust, drugs, cocaine, smoking;
- bacteremia;
- the presence of endoxins in the blood;
- exposure to volatile hydrocarbons;
- infections of various natures.
Also, many clinicians are inclined to believe that the disease is genetic. People with certain types of human leukocyte antigens are more susceptible to this disease and have a worse prognosis for its course.
Perhaps this is the reason why goodpasture syndrome most often occurs in white men aged 20 to 30 years, and then in women in the older age group - over 60 years. Although, in general, the disease is quite rare, affecting 0.5-1.8% of people per million per year.
There are several main options for the development of pathology:
- Rapidly progressing glomerulonephritis and hemorrhagic pneumonia, prone to relapse.
- Slow progression of changes in target organs.
- Primary kidney damage with the development of renal failure.
As mentioned earlier, the causes of the occurrence have not yet been precisely established. There are several possible factors for the development of the syndrome:
- Predisposition at the genetic level.
- Consequences of past infections of both bacterial and viral nature.
- Taking certain medications (penicillamine drugs).
- Prolonged hypothermia.
To this day, the pathogenesis and etiology of Goodpasture's syndrome have not been fully elucidated. Many medical scientists note a person’s hereditary predisposition to the disease.
The following factors provoke the occurrence of pathology:
- Taking certain groups of medications.
- Hypothermia.
- Alcohol abuse.
- Work in hazardous industrial conditions.
- Oncopathology.
- Smoking.
- Acute infectious diseases.
- HIV AIDS.
- Intoxication due to the action of chemicals,
- Addiction.
Affected Populations
Goodpasture syndrome is a rare autoimmune disease that appears to affect men more often than women. The age of onset is usually between 20 and 30 years, but people of any age can be affected.
The disease was first identified in 1919. Since then, approximately 600 cases have been reported in the medical literature.
In Russia, anti-GBM disease is a rare disease; approximately 1-2% of all cases of rapidly progressive glomerulonephritis are secondary to this disorder.
Goodpasture's syndrome in children
Information that the disease affects only experienced smokers is unreliable, since there are cases of infection in children. Most often, the disease affects adolescents over 16 years of age after suffering severe infectious or bacterial infections. This process creates cross-antibodies that attack microbes and body tissues equally.
Goodpasture syndrome is a rapidly progressing pathological process that is dangerous if it was diagnosed already in pulmonary failure. The disease has a poor prognosis, since there is always a possibility of spontaneous relapse and a low survival rate for pulmonary hemorrhages. Timely x-ray checks will help diagnose the disease in time and give you a long, happy life.
| Procedures and operations | average price |
| Rheumatology / Consultations in rheumatology | from 600 rub. 297 addresses |
| Urology / Diagnostics in urology / Ultrasound in urology | from 100 rub. 1058 addresses |
| Cardiology / Diagnostics in cardiology / EPI in cardiology | from 100 rub. 1009 addresses |
| Cardiology / Diagnostics in cardiology / Ultrasound of the heart | from 300 rub. 763 addresses |
| Pulmonology / Diagnostics in pulmonology / Study of respiratory function | from 250 rub. 474 addresses |
| Pulmonology / Consultations in pulmonology | from 563 rub. 245 addresses |
| Pulmonology / Diagnostics in pulmonology / Radiography in pulmonology | from 350 rub. 180 addresses |
| Urology / Consultations in urology | from 563 rub. 128 addresses |
| Urology / Kidney surgeries / Kidney resections | RUB 104,775 90 addresses |
| Urology / Diagnostics in urology / Biopsy in urology | from 1650 rub. 82 addresses |
Diagnosis of the syndrome
In the diagnostic study of pathology, and when a differential diagnosis is made, taking an anamnesis is very important, especially along the hereditary genetic line.
Diagnostics begins with testing the blood composition to detect DNA antibodies - using the enzyme immunoassay method.
For a more complete picture, it is necessary to undergo laboratory and instrumental studies of the disease:
- General blood analysis;
- General urine analysis;
- Immunogram method;
- Sputum analysis;
- Microscopy of tissue cells of the kidney organ, as well as lungs;
- X-ray;
- Measuring respiratory rate and volume of air inhaled by the lungs;
- ECG (electrocardiography of the heart organ);
- Spirography method;
- CT scan of the lungs (computed tomography);
- Measuring the pressure in the blood of the arteries of oxygen ions and carbon dioxide molecules;
- Ultrasound (ultrasound examination) of the kidneys;
- ECHO-cardiography of the heart.
During diagnostic study, it is necessary to clearly distinguish Goodpasture syndrome from:
- Diseases: Wegener's granulomatosis;
- Polyarteritis nodular type;
- Systemic lupus erythematosus;
- Hemorrhagic type of vasculitis.
These pathologies have an autoimmune etiology and similar symptoms, but they lack antibodies that manifest themselves against antigens.
Diagnostic criteria
It is quite difficult to diagnose Goodpasture syndrome and establish an accurate diagnosis, since many signs of the pathology are nonspecific and the symptoms are very similar to many other diseases.
For this reason, criteria for the diagnostic study of renal-pulmonary syndrome have been established:
- A combination of signs of lung bleeding with kidney pathology - glomerulonephritis. This is the first sign of Goodpasture syndrome;
- Progression of pathology and rapid development of renal organ failure and respiratory system failure;
- Anemia, which manifests itself against the background of a deficiency of iron molecules in the body;
- Increased concentration of antibodies in the blood that show aggression to the basement membrane of the pulmonary alveoli, as well as to the glomeruli of the kidneys;
- Antibody components are accompanied by capillaries.
Diagnosis is carried out very carefully and it is necessary to exclude pathologies that also manifest themselves as hemoptysis:
- Pulmonary tuberculosis;
- Neoplasms in the lungs;
- Oncological tumors in the bronchi;
- Abscess of tissue of the pulmonary organ;
- Pneumosclerosis with bronchial ecstasies;
- Pathology of the cardiac organ, combined heart defects that develop hypertension of pulmonary etiology;
- Pathologies of the blood flow system;
- Diseases that are included in the group of systemic vasculitis;
- Diathesis of hemorrhagic manifestations.
What is Goodpasture syndrome
Goodpasture syndrome is a severe pathology, which is an autoimmune syndrome with pulmonary hemorrhage and glomerulonephritis. This is a systemic capillaritis with predominant damage to the lungs and kidneys, such as hemorrhagic pneumonitis and glomerulonephritis. Men aged 20 to 30 years are more likely to get sick. According to medical observations, Goodpasture syndrome most often develops in people with a hereditary predisposition who smoke cigarettes. Additional negative factors are viral infections, inhalation of hydrocarbons and some other factors. There is a connection with viral and bacterial infection, hypothermia.
The first description of the disease was made during the influenza epidemic in 1919. Autoimmune pathogenesis is discussed, since circulating and fixed antibodies to the basement membranes of the kidneys were found, cross-reacting with antigens of the basement membranes of the lungs.
One of the doctor’s significant achievements is the development of a new method for growing viruses on chicken embryos and eggs. This technology has made it possible to develop vaccines against influenza, chickenpox, smallpox, yellow fever, typhus, Rocky Mountain spotted fever and some other viral pathologies.
For outstanding achievements in science and medicine, Ernst William Goodpasture was awarded a Rockefeller Foundation Fellowship, the John Scott Medal, the Kober Medal and a number of other prestigious awards and honors. He received honorary doctorates from Yale University, the University of Washington, Tulane University and the University of Chicago.
Prevention recommendations
Since the exact pathogenesis of the disease is currently unknown, no algorithm for its prevention has been developed. General principles boil down to timely consultation with a doctor. Although the syndrome is characterized by rapid development, it is much easier to combat it in the early stages of its occurrence. Since the triggers for the formation of the disease are viral and bacterial infections, preventive recommendations include avoiding infection. To do this, you need to adhere to the principles of a healthy lifestyle, which means proper and balanced nutrition, moderate physical activity and giving up bad habits. This approach supports the natural functioning of the immune system, being the main condition for preventing the problem.
Goodpasture's syndrome
Goodpasture's syndrome (synonyms: hemorrhagic pneumonia with nephritis, idiopathic pulmonary hemosiderosis with nephritis, pulmonary hemorrhage with glomerulonephritis, persistent hemophthisis with glomerulonephritis) is a pathological process based on autoimmune disorders with damage to the basement membranes of the pulmonary alveoli and renal glomeruli. The clinical picture was first described in 1919 by E. Goodpasture. Goodpasture syndrome is a rare disease; most often affects young people (from 12 to 35 years). Among the patients, men predominate.
The etiology of the disease is unknown. Its occurrence is sometimes associated with inhalation of volatile hydrocarbons and other organic solvents. A number of authors consider Goodpasture syndrome as a variant of the course of idiopathic pulmonary hemosiderosis, which is indirectly confirmed by the existence of clinically and pathomorphologically transitional states. Unlike collagenosis, with Goodpasture syndrome there are no symptoms of vasculitis. The assumption of a viral nature does not have sufficient factual evidence.
Pathogenesis. It is now considered proven that patients with Goodpasture syndrome develop autoantibodies to the basement membranes of the pulmonary alveoli and renal glomeruli (which have common antigenic structures). Several hypotheses have been expressed about the reason for the formation of these autoantibodies - under the influence of exogenous antigens, antibodies are formed that damage the alveolar and glomerular basement membranes; changes in basement membranes occur as a result of infection or exposure to toxic chemical factors (organic solvents, etc.); impairment of the excretory function of the kidneys slows down the excretion of breakdown products of basement membranes, which leads to the formation of autoantibodies to them.
The question of the primary localization of the disease has not been completely resolved, although lung damage, as a rule, precedes kidney damage.
Pathological anatomy. Histological examination reveals hemorrhagic necrotizing alveolitis (changes in the lungs similar to idiomatic pulmonary hemosiderosis) and nephrosonephritis with focal intracapillary thrombotic changes and the development of glomerular fibrosis.
Clinic. The most characteristic signs of Goodpasture syndrome are hemoptysis (repeated pulmonary hemorrhages) and anemia. They are often accompanied by progressive shortness of breath, cough, chest pain, and weight loss. Radiographs reveal finely reticulated deformation of the pulmonary pattern and small focal changes. Body temperature may rise.
To these, as a rule, the first signs of Goodpasture's syndrome, symptoms of kidney damage are soon added: proteinuria, hematuria, cylindruria, increased levels of residual nitrogen in the blood, peripheral edema. Hypochromic iron deficiency anemia progresses. When a secondary infection occurs, corresponding changes in the blood count are noted.
The course of the disease is steadily progressive and ends in death due to repeated pulmonary hemorrhages or azotemic uremia.
Treatment. Early administration of corticosteroids and immunosuppressants (azathioprine, cyclophosphamide, 6-mercaptopurine, etc.) can slow down the progression of the process. Repeated hemodialysis slightly prolongs the life of patients. For intractable uremia (after preliminary hemodialysis), nephrectomy (followed by kidney transplantation) is sometimes performed, which eliminates the source of antigenic irritation. In recent years, plasmapheresis has been used to remove circulating antibodies.
It has been noted that antibodies against the basement membranes of the renal glomeruli cease to be detected in the blood serum after 6-8 months from the onset of the disease. Therefore, intensive therapy during this period (corticosteroids, immunosuppressants, hemodialysis, plasmapheresis, nephrectomy) may help prolong the patient’s life until autoantigenogenesis stops.
Symptomatic therapy includes repeated blood transfusions and the administration of iron supplements.
The prognosis is unfavorable.
Signs and symptoms
The main symptoms of Goodpasture syndrome are excessive bleeding into the lungs (pulmonary hemorrhage) and inflammation of the filtering structures (glomeruli) or kidneys (glomerulonephritis). In some cases, an upper respiratory tract infection may precede the development of the disorder. Common symptoms associated with the syndrome may include:
- fever;
- nausea;
- fatigue.
Pulmonary hemorrhage can lead to episodes in which patients cough up blood (hemoptysis). The severity of this sign can range from a few spots to an excessive amount of blood. Those affected may also have difficulty breathing (shortness of breath), lethargy, chest pain, a dry, rough sound from the throat, and/or a frequent cough. In rare cases, those affected may experience an abnormal accumulation of fluid (edema) in the lung tissue. Pulmonary abnormalities are usually present before or simultaneously with renal abnormalities in about 70 percent of cases.
Inflammation of the filtering structures (glomeruli) of the kidneys (glomerulonephritis) can cause the kidneys to be unable to process waste from the blood and eliminate it in the urine (acute kidney failure). Kidney failure usually results in a decrease in the amount of urine the body produces. Additional symptoms associated with kidney failure may include:
- pale skin;
- drowsiness;
- nausea and/or vomiting.
Severe complications of kidney failure include bleeding in the stomach and/or a decrease in the number of circulating red blood cells (anemia).
In rare cases, victims may experience high blood pressure (hypertension) and/or joint pain and swelling (arthritis). In some cases, symptoms of Goodpasture syndrome may recur after treatment.
Reasons for development
The etiology of the disease still remains unclear. It is believed that the pathological process is based on immunological damage: antibodies are produced to the basement membranes of the renal glomeruli and pulmonary alveoli, which lead to inflammation and disruption of the kidneys and lungs. The combined damage to these organs is explained by the common antigenic structure of their basement membranes.
It is not uncommon for the syndrome to occur shortly after influenza or inhalation of hydrocarbons. In this regard, it is assumed that such influences change the chemical structure of membrane antigens. As a result, they acquire autoantigenic properties, causing the production of antibodies. Thus, regarding the reasons for the formation of autoantibodies, there are several hypotheses:
- infectious diseases or toxic effects of chemicals;
- the action of exogenous (entered the body from the environment) antigens;
- slowing down the excretion of breakdown products of basement membranes due to impaired excretory function of the kidneys.
The exact cause of Goodpacher's syndrome has not yet been established. The most studied cases are related to:
- viral infection (usually influenza, hepatitis A virus);
- medications (antibiotics of the penicillin group, Carbimazole);
- harmful and toxic substances in production (gasoline fumes, solvents, paint and varnish and tobacco production);
- significant hypothermia of the body;
- chronic nicotine poisoning from smoking.
A genetic predisposition has been identified in individuals who have specific alleles (regions) in genes (HLA-DRwl5, HLA-DR4 and HLA-DRB1). The possibility of hereditary transmission has been confirmed by cases of Goodpasture syndrome in individuals from the same family.
Causes
The cause of Goodpasture syndrome is unknown. Therefore, measures to prevent it have not been developed.
Some factors have been identified that may be related to the occurrence of the disease:
- viral infections, such as influenza A2c,
- prolonged contact with gasoline and organic solvents,
- smoking,
- shock wave lithotripsy (“crushing stones”) in the ureter for urolithiasis,
- genetic characteristics - the presence of certain genes of the HLA system in a person.
Acute onset: symptoms of Goodpasture syndrome
The onset is acute with high fever, hemoptysis or pulmonary hemorrhage, and shortness of breath. On auscultation there is an abundance of sonorous moist rales in the middle and lower parts of the lungs, and radiologically there are multiple focal or confluent darkening in both pulmonary fields. Severe, progressive glomerulonephritis develops almost simultaneously, quickly leading to renal failure. Repeated hemoptysis and hematuria lead, as a rule, to anemia, which is aggravated by renal failure. Laboratory examination: anemia, white blood cells and increased ESR. A characteristic immunological sign of the disease is antibodies to the basement membranes of the kidney. The prognosis is usually unfavorable - death occurs in the next 6 months - 1 year from the onset of the disease with symptoms of pulmonary-cardiac or renal failure.
In clinical practice, it is customary to distinguish three variants of the course of Goodpasture syndrome: malignant, moderate and slow.
- A malignant form of Goodpasture's syndrome. In the malignant form, recurrent hemorrhagic pneumonia is observed. In this case, glomerulonephritis develops rapidly. The malignant form of the pathology begins with pulmonary hemorrhages and acute renal failure, which requires intensive therapy.
- Moderate form. In this case, pulmonary-renal syndrome develops slowly and is quite moderately expressed.
- Slow form. In this case, the clinical picture is dominated by the phenomena of glomerulonephritis and chronic renal failure, but pulmonary manifestations of the disease develop quite rarely.
Of the specific symptoms of Goodpasture syndrome, the lungs are usually the first to be affected; The patient has a cough, progressive shortness of breath, chest pain, hemoptysis and pulmonary hemorrhage. Often, Goodpasture syndrome is accompanied by complications in the form of cardiac asthma and pulmonary edema.
After a certain time, the pulmonary manifestations of the syndrome are accompanied by renal symptoms: oliguria, hematuria, arterial hypertension, peripheral edema and others. In some cases, Goodpasture's syndrome is accompanied by arthralgia, myalgia, skin hemorrhages, pericarditis and some other symptoms.
Types of pathology
According to the clinical course of Goodpasture syndrome, there are 3 main types:
- malignant syndrome with often recurrent pneumonia with severe course and rapidly progressive glomerulonephritis;
- with slow and less pronounced changes in the paired respiratory and excretory organs;
- syndrome with a predominant intensity of development of renal failure.
Medical news
At a working meeting in the government of St. Petersburg, it was decided to more actively develop the HIV prevention program. One of the points is: testing for HIV infection up to 24% of the population in 2020.
Experts agree that it is necessary to attract public attention to the problems of cardiovascular diseases. Some are rare, progressive and difficult to diagnose. These include, for example, transthyretin amyloid cardiomyopathy
On October 12, 13 and 14, Russia is hosting a large-scale social event for free blood clotting testing - “INR Day”. The campaign is timed to coincide with World Thrombosis Day.
The incidence of meningococcal infection in the Russian Federation in 2020 (compared to 2020) increased by 10% (1). One of the common ways to prevent infectious diseases is vaccination. Modern conjugate vaccines are aimed at preventing the occurrence of meningococcal infection and meningococcal meningitis in children (even very young children), adolescents and adults.
The long weekend is coming, and many Russians will go on holiday outside the city. It's a good idea to know how to protect yourself from tick bites. The temperature regime in May contributes to the activation of dangerous insects...
How it develops
This disease is autoimmune. Antibodies appear in the blood directed against the glomerular membrane in the kidneys. They bind to sensitive membrane cell receptors and trigger a chain of reactions leading to tissue damage.
Mechanism of autoimmune damage to the vascular wall
The basement membrane is composed of collagen and in turn is a supporting structure for the inner and outer vascular epithelium, and they are ubiquitous in the human body. But only in the alveoli of the lungs and in the glomeruli of the kidneys do the basement membrane cells suffer clinically, since they contain a larger number of structures that the immune system recognizes.
In addition, since the entire volume of blood constantly passes through the kidneys, glomerular cells are more accessible to circulating antibodies.